This exercise heps you to understand VSEPR model and orbital hybridization concept to predict molecular shapes.
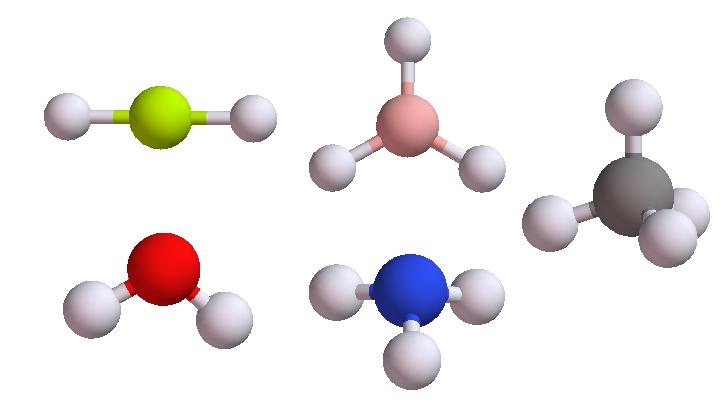
Part A: Using the VSEPR model to predict the structures for BeH2, H2O, BH3, NH3, and CH4 and the angles of the central atoms.
Part B: Use Basic QChem Edu or Basic QChem tools in Avisto to find the stable structures for the above molecules. You can download these tools from Astonis.
Procedure:
- Use MolDesign to create BeH2, H2O, BH3, NH3 and CH4 then send them to your workbench.
- Use Basic QChem Edu or Basic QChem tool to search for stable structures of these molecules.
- Use PsiViewer to analyze the results (just double-click on the data sent out in Step 2).
- Measure the angle of the central atom by click on the atoms defining the angle.
- In the MO tab, select to plot the delocalized orbitals. Classify these orbitals as bonding, anti-bonding, or non-bonding.
- In the MO tab, select to plot the localized orbitals. For each molecule, plot all localized orbitals and notice the size of the electron lone pairs on H2O and NH3.
- On the MO coefficients panel, select not to plot contributions from the Hydrogen atoms, then replot to view the hybrid orbitals of the central atom.