In the Linear Combination of Atomic Orbitals (LCAO) approximation, a molecular orbital of a molecule can be written as
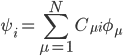
where N is the total number of atomic orbitals from its constituent atoms and 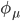
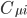
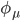
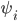
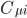
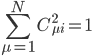
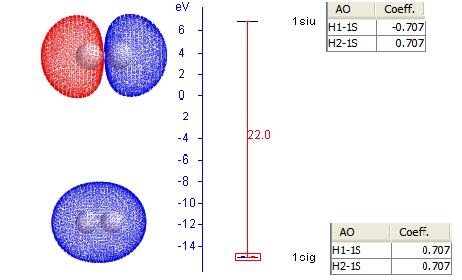
Using H2 for example, each hydrogen atom has one 1s atomic orbitals. With two AO's, we have two MO's. The results of LCAO are shown below on the orbital energy diagram with the MO coefficients. This figure is composed from plots in PsiViewer in Avisto.
Generalize this concept to homonuclear diatomic N2 molecule, the resulted MO's from the LCAO approximation is plotted on its orbital energy diagram using the same tool.
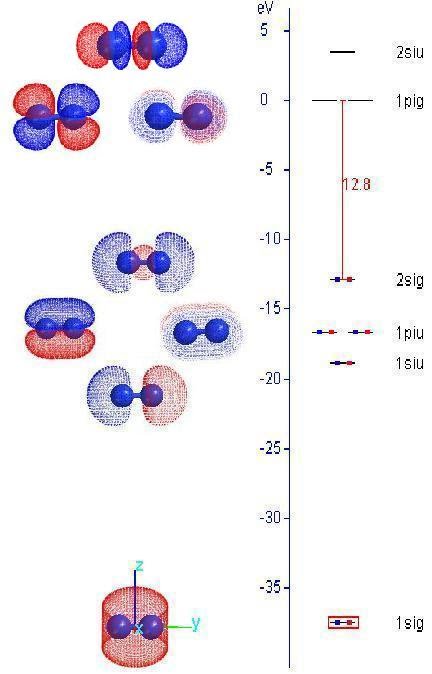
Repeat the experiment for N2 and notice the MO coefficients for each MO.
At this point, you can repeat the calculations for the remaining of
homonuclear diatomic molecules from atoms in the second row of the periodic table, namely Li2, Be2, B2, C2, O2 and F2.
You can download tools for this experiment from Astonis.