In the Valence Bond model, atomic orbitals s and p can be mixed to yield a set of hybrid orbitals for forming sigma bonds with neighbor atoms for example of the sp3 and sp2 hybrid orbitals below.
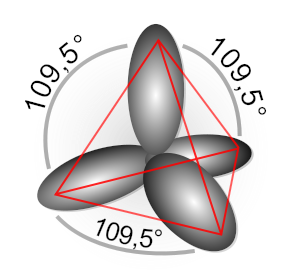
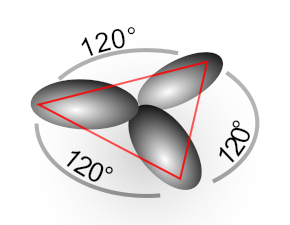
This provides a means to ensemble molecular structure from individual atoms such as in MolDesign tool in Avisto. Let examine the concept of orbital hybridization from the molecular orbital theory, i.e. analyzing molecular orbitals from semi-empirical MO calculations using tools in Avisto.
The procedure below is for using tools in Avisto. Dowload them at Astonis.
Procedure:
1. Use MolDesign to build BeH2, HCCH, BH3, H2C=CH2, CH4, and H3C-CH3.
2. Use Basic QChem Edu, Basic QChem, or MopacGUI Cloud or Pro to search for stable structures for these molecules. If use MopacGUI Cloud or Pro also select options to calculate localized MO and perform hybridization analysis under that properties tab.
3. Use PsiViewer to analyze the molecular orbitals both delocalized and localized forms.
4. Open the output file in the Files type to view the results of the hybridization analysis.
Example: BeH2
After using Basic QChem Edu to search for a stable structure of BeH2, view the results in PsiViewer.
1. Delocalized molecular orbtials
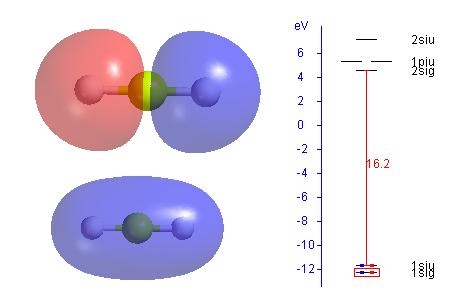
The figure shows both occupied delocalized 
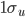
2. Select option to plot localized MO in PsiViewer. The two delocalized MO's above are mixed to produce two equivalent localized MO's showing the two Be-H sigma bonds.
Note the the orbital energies of localized MO's have no physical meaning as those of delocalized MO's.
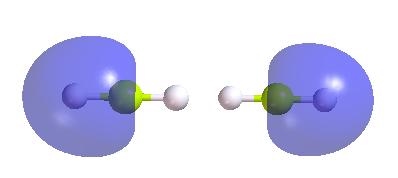
3. Using option in PsiViewer to turn-off the contribution of Hydrogen atoms in the localized MO's. This yields two Be hybrid sp orbitals.
4. Open the output file in the Files tab of PsiViewer and find the table 'Sigma-Pi bond-order matrix'
SIGMA-PI BOND-ORDER MATRIX
S-SIGMA P-SIGMA P-PI S-SIGMA S-SIGMA Be 1 Be 1 Be 1 H 2 H 3------------------------------------------------------------------ S-SIGMA Be 1 0.996282 P-SIGMA Be 1 0.000000 0.961062 P-PI Be 1 0.000000 0.000000 0.000000 S-SIGMA H 2 0.498141 0.480531 0.000000 0.983320 S-SIGMA H 3 0.498141 0.480531 0.000000 0.004648 0.983320
Along the diagonal matrix, the first two numbers indicate that Be makes two sigma bonds from an s and p orbitals and no pi bond. This also gives the degree of orbital hybridization to be a sp type.
You can repeat the lesson for other molecules to learn about sp2 and sp3 hybridization.