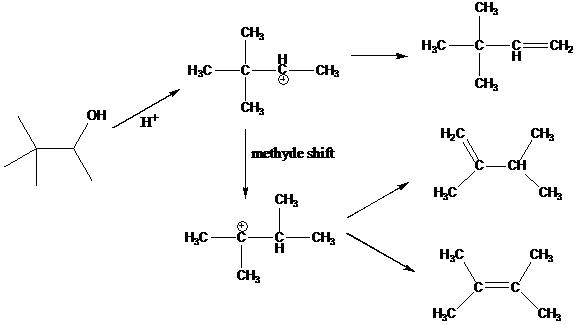
Heating alcohols in a strong acid leads causes it to dehydrate and form an alkene. However, for some primary and secondary alcohols, rearrangement occurs during the dehyration process that leads to formation of different alkenes. It is known that such dehydration reaction in general favors the production of the more stable alkene.
This experiment is to examine the relative stability of possible alkene products from dehydration of 3,3 dimethyl 2-butanol to explain for the observation of 80% of 2,3-Dimethyl-2-butene, 20% of 2,3-Dimethyl-1-butene, and a trace of 3,3-Dimethyl-1-butene. The mechanism for this dehydration is shown below.
Procedure: Using tools in Avisto. You can download Avisto and its tools from Astonis.
- Use MolDesign to build the three alkenes 2,3-Dimethyl-2-butene, 2,3-Dimethyl-1-butene, and 3,3-Dimethyl-1-buteneas as shown in the figure above.
- Use Basic QChem Edu, Basic QChem, Mopac GUI Cloud or Pro to search for stable structures of the three alkenes.
- Compare the relative heats of formation and rank them from the lowest heats of formation which corresponds to the most stable alkene to the least.
- Is your ranking consistent with what observed experimentally for the production distribution?
Related problem:
Using the above analysis to predict the product distribution for dehydration of these two bicyclic alcohols
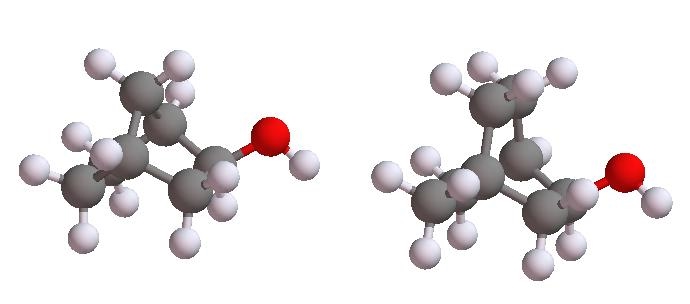
Does your results confirm Brendt's rule that the dehyration process leads to formation of double bond away from the bridgehead carbon atoms?