Here is the description of pi-backbonding from Wikipedia.
π backbonding, also called π backdonation, is a concept from chemistry in which electrons move from an atomic orbital on one atom to a π* anti-bonding orbital on another atom or ligand. It is especially common in the organometallic chemistry of transition metals with multi-atomic ligands such as carbon monoxide, ethylene or the nitrosonium cation. Electrons from the metal are used to bond to the ligand, in the process relieving the metal of excess negative charge. IUPAC offers the following definition for backbonding:
A description of the bonding of π-conjugated ligands to a transition metal which involves a synergic process with donation of electrons from the filled π-orbital or lone electron pair orbital of the ligand into an empty orbital of the metal (donor–acceptor bond), together with release (back donation) of electrons from an nd orbital of the metal (which is of π-symmetry with respect to the metal–ligand axis) into the empty π*-antibonding orbital of the ligand.
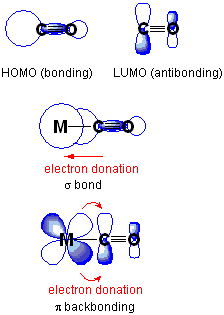
The electrons are taken from a d-orbital of the metal, and usually placed into an anti-bonding molecular orbital of the ligand, which causes the bond order of that ligand to decrease. This leads to a decrease in the vibrational frequency of the bond, which can be measured using IR spectroscopy. Although the bond-order within the ligand is decreased by this process, the metal-ligand bond order is increased.
This experiment is to demonstrate the pi-backbonding concept using molecular orbital theory for several transition metal complexes, namely Co(CO)6, Fe(CO)5, and Ni(CO)4 and to confirm the effects of pi-backbonding on the bond order and vibrational frequency of CO.
Procedure: Using tools in Avisto. You can download Avisto and its tools at Astonis.
- Use MolDesign to create CO, and the three
Co(CO)6, Fe(CO)5, and Ni(CO)4 complexes.
- Use Basic Qchem, Mopac GUI Cloud or Pro to search for stable structures of CO and of these complexes.
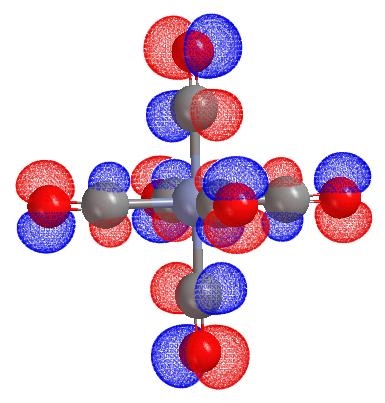
Open the Co(CO)6 result and plot the HOMO orbital. You will be able to view the d-pi orbital as shown below.

by select to remove the contributions of Co d orbitals in this HOMO orbital, then replot. You will obtain the pi* orbitals of all CO ligands.
You can also view the sigma-electron donation from the CO ligands in the HOMO-4 orbital.

Repeat the experiment for other complexes.
- Record the bond length of isolated CO and CO in different complexes with their bond orders. To obtain the bond order information, you need to open the .out file in the File tab and look for the BOND ORDERS AND VALENCIES. Locate the element between two atoms that you are looking for. For example, for the Co(CO)6 complex, the C=O bond orders for all ligands are about 2.13 compared to 2.37 in isolated CO. This indicates the CO bond is weaken due to pi-backbonding. The CO bond length in the ligans are also somewhat longer compared to that of isolated CO as well.
- Perform vibrational analyses for all species. Compare the CO strectching frequency in isolated CO with those of CO ligands in the complexes.